AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Atom vs molecule10/11/2023 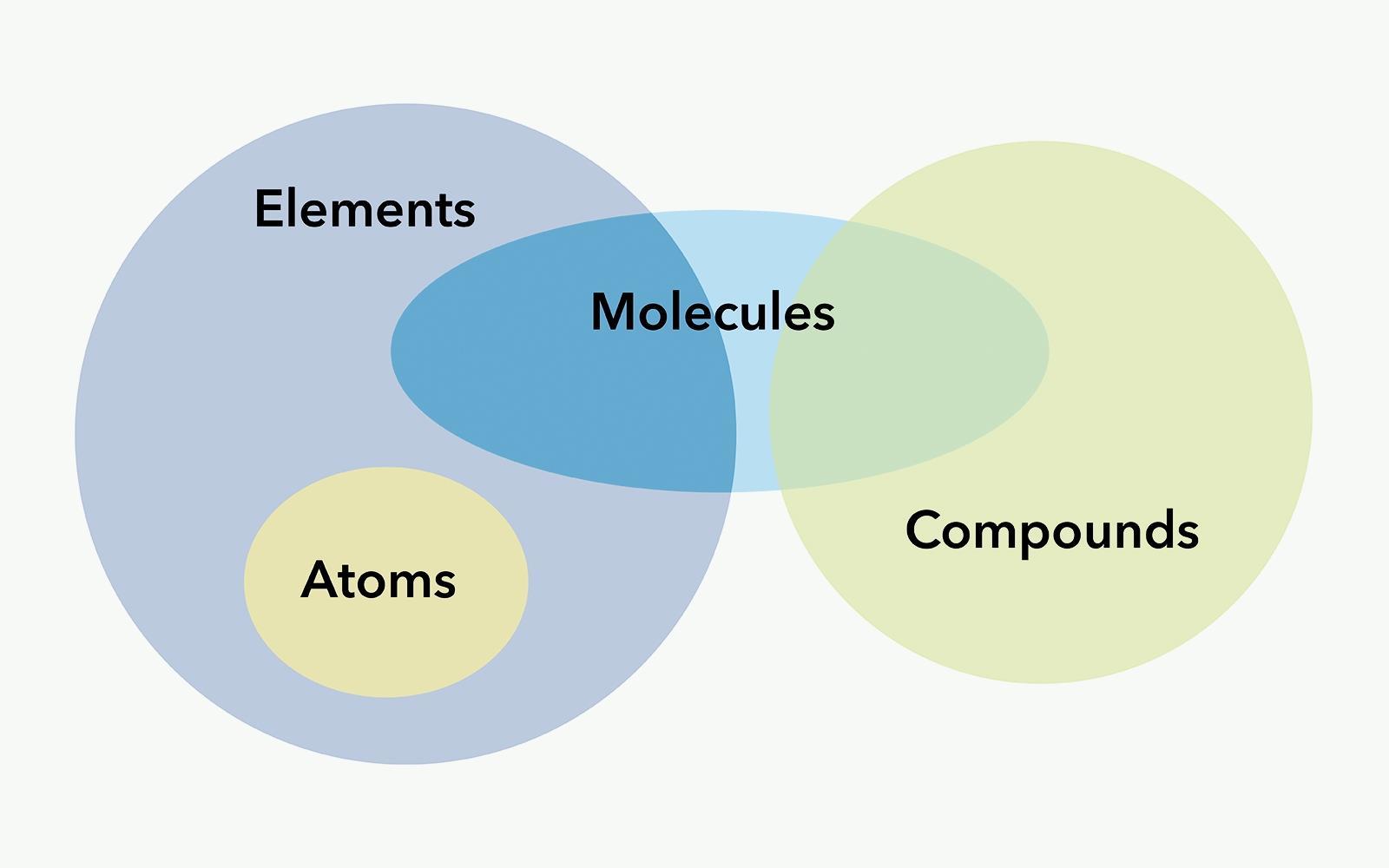
The components of the atom are held together by means of nuclear bonds. Molecules are made of atoms of the same or different elements. Molecules are the basic unit of a compound.Ītoms are made up of Protons, Neutrons and Electrons. It is expressed in Atomic Mass Unit (amu). It is roughly equivalent to the mass of the total protons and neutrons in the nucleus as the mass of electrons is negligible. Atomic Mass: It is the total mass of the atom in an element.Mass number: Mass number is the total number of protons and neutrons present in the nucleus of the atom.Atomic number: Atomic number is the total number of protons present in an atom.The number of protons in an atom of an element remains constant whereas the number of neutrons can change. Neutrons: Neutrons are neutral particles present inside the nucleus of the atom.The mass of the electrons is 1/1836 the mass of protons, thus making it negligible. The electrons orbit around the nucleus in a series of levels called energy levels. Electrons: Electrons are negatively charged particles that revolve around the nucleus. 
Protons along with neutrons form the nucleus and together they are called the nucleons. 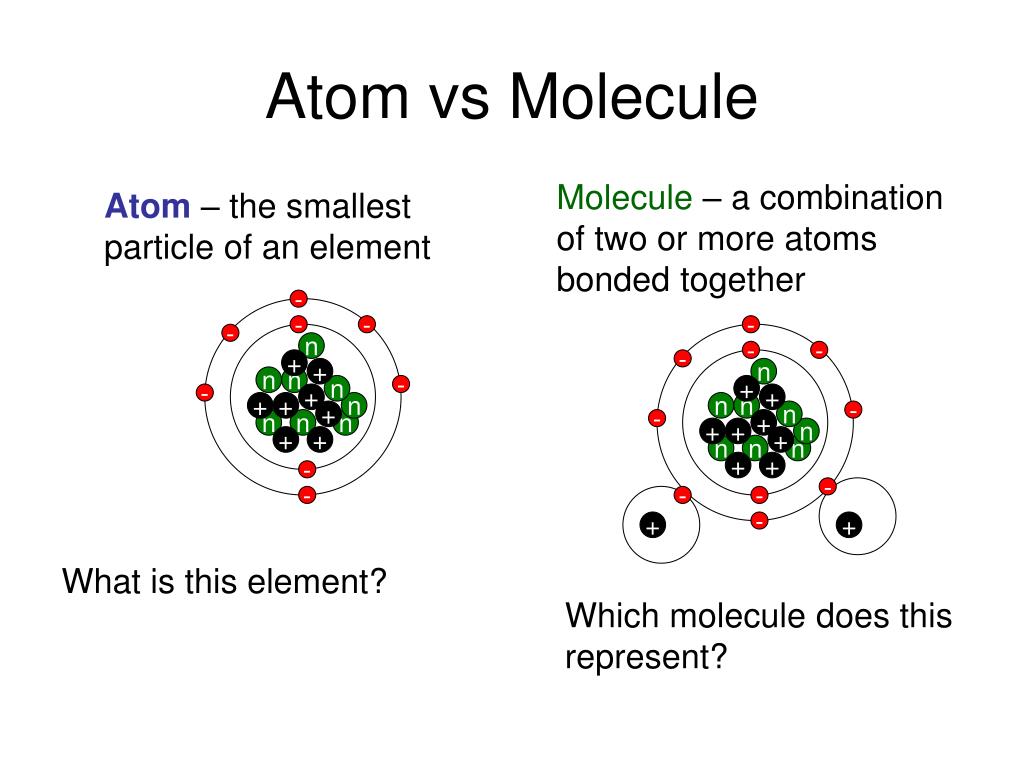
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
